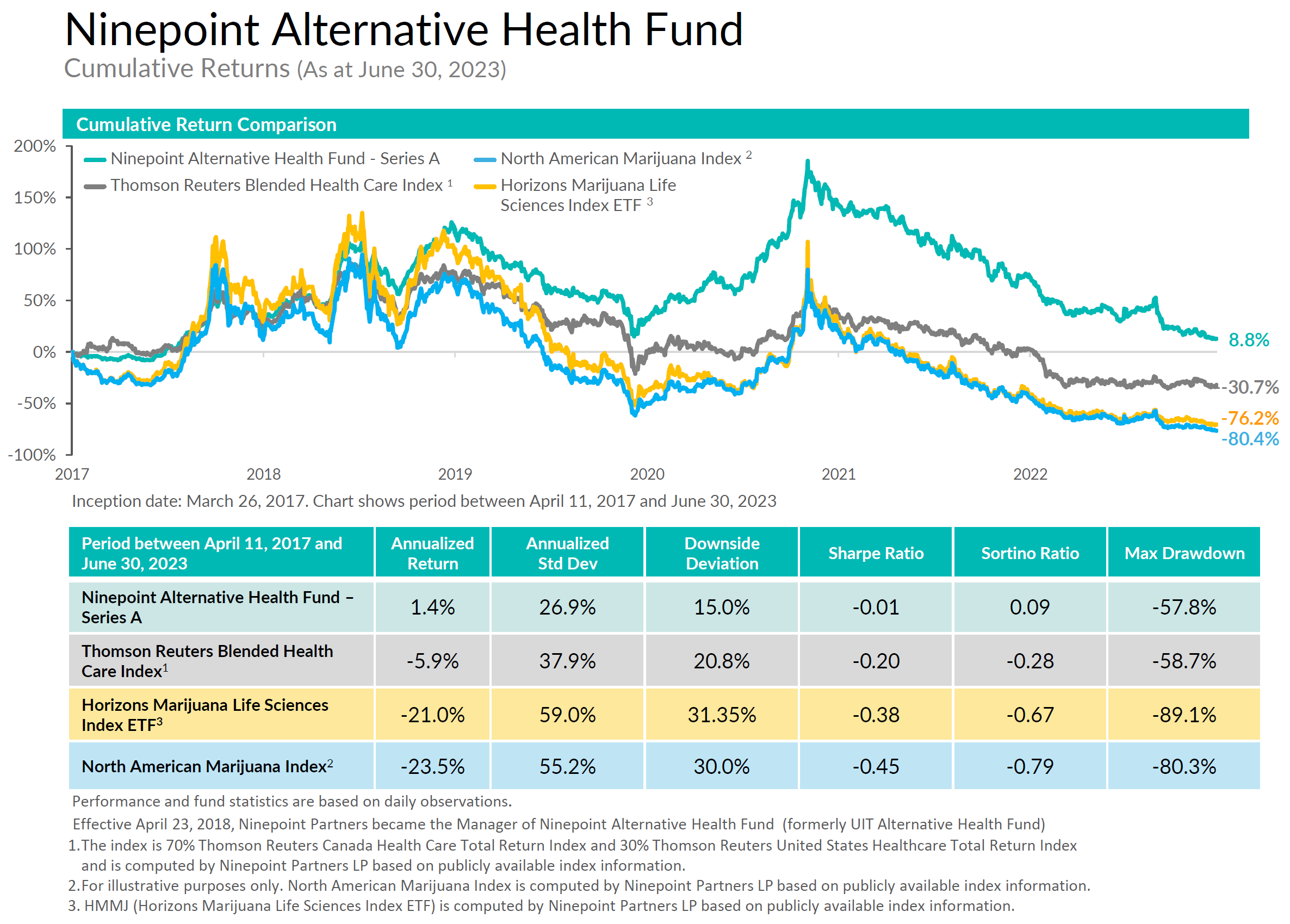
Ninepoint Alternative Health Fund
June 2023 Commentary
As we have reached the mid-point in 2023, it is important to look at what is driving equity prices within our mandate of the Ninepoint Alternative Health Fund. Healthcare related company performance has been muted despite the demand for medical services, elective surgeries and various pharmaceutical solutions. Post pandemic, we have adjusted select names in healthcare and pharma that reflect ongoing and growing demand. Most healthcare related businesses in Q1-Q2 have maintained or increased guidance for 2023 however equity market participants have largely ignored that strength in favour of investing in select large cap tech stocks offering the future potential of AI. With valuations for these large cap tech stocks at significant multiples of sales, we believe that investing in those companies that are exhibiting current growth in demand and cash flow growth at low multiples are preferred choices. On the US cannabis front, the lack of federal regulatory announcements has resulted in investor apathy for the sector despite the strong cash flows being generated by the leading US cannabis companies. Further weighing on cannabis and consumer health companies is that consumers continue to worry about interest rates, inflation and the fear of recession. These factors result in higher costs for goods and services and less cash in consumer pockets as mortgages and credit card debt continue to rise. In the second half of 2023 we look to more buoyant returns for healthcare and consumer health as demand is maintained in this challenging environment, while other sectors begin to weaken under the strain of the economic realities faced by global markets.
The month of June had positive contributions from healthcare and cannabis names as several top Fund positions contributed to the fund’s return. Leading contributors for the Fund included US Cannabis company Terrascend (TSX:TSND) +7.21%; COSTCO COST +5.24%; Eli Lilly LLY +9.20%; Kenvue KVUE 5.30%; Abbott Labs ABT +6.88%; Johnson & Johnson JNJ +6.75%.
In this month’s commentary, we focus on the recent announcements from biotech company Eupraxia (EPRX) as it announced very strong phase 2b clinical trial results with respect to its pain management solution in the treatment of knee osteoarthritis patients. We also discuss US cannabis announcements from the US Dept of Health & Human Services with HHS Secretary Becarra discussing 2023 delivery of President Biden’s re-scheduling plans for cannabis within the Controlled Substances Act (CSA). Also noteworthy in US cannabis is the opening of the state of Maryland as a new Rec state effective July 1st and the potential growth that well placed participants can achieve. On the US cannabis side, we also highlight a Top Ten Fund holding, TSND on its TSX uplisting efforts as well as activity in building its operations in MD ahead of the July 1st recreational market opening. Finally, we highlight the biotech acquisition of Dice Therapeutics (DICE) by Eli Lilly & Co (LLY) which focuses on auto immune therapies.
Eupraxia: Strong Phase 2b Trial Results
A pharmaceutical company the Fund invested in over 20 months ago on the basis of a compelling clinical trial focused on improving the pain management and safety of treating osteoarthritis patients based on a slow release reformulation of an existing approved drug. Using an existing drug substantially decreases risks in clinical trials. Eupraxia Pharmaceuticals (EPRX) released very strong top line data from their Phase 2b pain trial that enrolled a total of 318 moderate to severe osteoarthritis patients being treated for knee pain related to OA.
According to Precedence Research, the osteoarthritis therapeutics market size accounted for USD 8.21 billion in 2022 and it is expected to hit around USD 18.36 billion by 2032, expanding at a CAGR of 8.38% from 2023 to 2032. Knee OA accounts for ~80% of the total OA therapeutics market in the US and close to 70% of patients have bilateral disease, meaning both knees exhibit pain that require treatment.
Current treatment protocols, such as non-steroidal anti-inflammatory drugs (NSAIDs) have been shown to be less than effective at maintaining relief from OA pain in addition to being linked to severe side effects. More than 50% of OA patients use prescription NSAIDs to control pain, which can have serious side effects; NSAIDs result in 41% of cardiovascular events in OA patients while up to 10% have an annual risk of gastrointestinal bleeding and up to 5% of NSAID users develop kidney problems.

The EPRX trial, therefore, is significant not just to find an alternative that provides longer lasting pain relief but also shows limited to no severe side effects. The Phase 2b trial was a double-blind, placebo-controlled study that generated results that showed the primary goal was met with a statistically significant reduction of pain in the knee as measured by the WOMAC pain score at 12 weeks in addition to showing significantly better than average pain relief than placebo at 24 weeks. WOMAC is a global standard test that examines pain management and drug effectiveness; The Western Ontario and McMaster Universities Arthritis Index (WOMAC) is widely used in the evaluation of Hip and Knee Osteoarthritis globally. In addition to meeting its primary goal, 3 of 4 secondary goals and all safety criteria were also met, demonstrating clinically meaningful and durable pain relief. EPRX drug has a safety and efficacy profile that could allow physicians to obtain treatment objectives that are beyond the scope of existing therapy and opens the door for the company to move to its Phase 3 development program, which will be supported by its Fast Track designation from the U.S. FDA. The company is also investigating other potential conditions amenable to treatment and has identified an encouraging candidate.
TerrAscend (TSND) Uplisting to the TSX
Top Ten fund holding TSND achieved several milestones during the month that have asserted it among the top securities to own in US cannabis. First, the company received conditional approval to list its equities on the Toronto Stock Exchange. Next, at the company AGM, investors overwhelmingly voted in favour of the uplisting move to the TSX. Finally, in the last week of June, the TSX approved the listing with July 4th being the first day of trading for TER to up list from the CSE to the TSX under the new ticker symbol TSND.
This is important as most US cannabis companies are listed on the Canadian Securities Exchange (CSE), considered by many US investors as not an exchange that provides necessary disclosure or liquidity to allow for positions to be maintained by Institutions. In addition, with the current informal regulatory guidance from US Custodians not to allow US cannabis equities to be held in their systems, the combined effect has resulted in muted liquidity, especially on the part of US institutional investors who have to date backed away from becoming meaningful investment participants. With equity securities being listed on the TSX, considered a senior North American exchange, it is estimated that the stigma related to the CSE could lift apprehension among US investors. Consider that TSX-listed banks, industrials and retailers (including Alimentation Couche Tard (ADT) are all widely held securities in the US. As a result, a TSX listing is believed to be a significant advantage to build a US investor following and enhance the daily liquidity volume and value of the company’s equity.
Terrascend (TSND) Announces Maryland Acquisitions
Early in the month, TSND signed a definitive agreement to acquire Peninsula Alternative Health an operator of a medical cannabis dispensary in Salisbury, Maryland. Peninsula is a leading dispensary in the state generating 2023 run-rate net revenues of $14.0mm, well above the MD state average of approx. $5 million. Peninsula could be one of TSND’s highest grossing stores in a REC market given limited competition (as there are no stores within 25 miles) and its location near the DE border, which could build sales due to the state’s Rec transition July 1st.
The purchase price is $22 million or 1.6x 2023 run-rate net sales, however, will only require a small cash outlay of $1.5 million. This is an important step in TSND’s strategy to go deep into select markets, where they have demonstrated leadership in cultivation and processing. Other state markets where their strategy is paying dividends includes New Jersey, Pennsylvania and Michigan. On top of the go-deep strategy, MD is going to be a strong state moving forward as the state goes rec July 1st.
Late in the month, TSND announced a further agreement to acquire Blue Ridge Wellness, another Maryland based cannabis dispensary, for ~US$6.75 million with US$3 million in cash. With current run-rate sales of US$4.3M (medical-only). TSND paid a transaction multiple of ~1.6x revenue resulting in the third MD dispensary acquisition by the company in 2023. TSND is keenly focused on the MD limited license nature of the market, combined with its Rec opening July 1st as it looks to reach the state-maximum of four retail locations as adult-use changes are implemented.
US Cannabis Re-Scheduling is a 2023 Decision
The US Secretary of Health and Human Services (HHS) Xavier Becarra announced that it is his intention to present President Joe Biden with a federal cannabis scheduling decision this year as agencies work to complete an administrative review. While Becerra and other federal officials have been working since the President's surprise announcement last October 6th, there’s been a lack of clarity about the specific timeline. Now the secretary has disclosed when he wants to deliver on the president’s directive. At the same time and under Secretary Becarra, the US Food and Drug Administration (FDA) is carrying out an eight-step scientific review into marijuana to determine whether it should be rescheduled, de-scheduled or remain in Schedule I, which is reserved for the most strictly controlled drugs under the Controlled Substances Act (CSA).
Becerra stated, “places like California have already changed the laws, the federal government has not, and so we’ve been instructed and we’re underway with that review as we speak.” Along with other agencies like the Drug Enforcement Administration (DEA), all agencies are working to give the president an answer that’s based on science and evidence.
The reason this is important is that many times stakeholders in the US cannabis industry have stated their desire to have marijuana/cannabis re-scheduled under the CSA from the strictest Schedule I narcotic it is listed as currently. This designation was done in the early 1970’s when US President Nixon, who deeply despised drugs, and had a deep paranoia of the hippie movement. These harsh policies had a disproportionate effect on visible minorities that over time has caused a significant amount of incarceration among various communities. Today, a majority of US voters believe that cannabis should be legalized at the federal level and the CSA designation review is a big step towards this goal.
LLY Acquires DICE To Build Immune Therapy Portfolio
Mid-month, Eli Lilly & Co (LLY) announced the acquisition of biotech company Dice Therapeutics (DICE) in a $2.4 billion deal to bolster its treatment portfolio for immune-related diseases. DICE is a biotech company that uses a proprietary technology platform to develop new oral therapeutic drugs for autoimmune diseases. Autoimmune diseases have a multitude of symptoms including pain, fatigue, dizziness, depression and rashes, with more than 100 known autoimmune diseases, including lupus, rheumatoid arthritis, Crohn’s disease and ulcerative colitis. LLY is using the strength in its share price, up 62% over 18 months as a way to acquire companies and treatment pipelines in order to bulk up its immunology business. Share price growth has been driven by its obesity drug tirzepatide, also known as Mounjaro, to drive future growth. LLY also has another development stage candidate in the obesity field called Retatrutide, which has generated some of the most impressive weight losses amongst this class of drugs so far.
DICE has an oral pill being tested in a mid-stage trial to treat chronic diseases related to auto immune deficiencies. This acquisition is a good complement to LLY's existing immunology franchise including Taltz, an LLY medication that treats plaque psoriasis and generated over $2.4 billion in revenues in 2022, while Olumiant, is a treatment for rheumatoid arthritis that generated $830 million in sales in 2022.
Option Strategy
Since the inception of the option writing program in September 2018, the Fund has generated significant income from options premium of approximately $4.79 million. We will continue to utilize our options program to look for attractive opportunities given the above average volatility in the sector as we strongly believe that option writing can continue to add incremental value going forward.
During the month we used our options strategy to assist in rebalancing the portfolio in favor of names we prefer while generating approximately $32,000 in options income. We continue to write covered calls on names we feel are range bound near term and from which we could receive above average premiums which included Bristol-Myers Squibb Co (BMY), Jazz Pharmaceuticals (JAZZ) and UnitedHealth Group Inc. (UNH). We also continue to write cash secured puts out of the money at strike prices that offered opportunities to increase our exposure, at more attractive prices, to names already in the Fund including Merck & Co. Inc. (MRK) and Eli Lilly & Co (LLY).
The Ninepoint Alternative Health Fund, launched in March of 2017 is Canada’s first actively managed mutual fund with a focus on the cannabis sector and remains open to new investors, available for purchase daily.
Charles Taerk & Douglas Waterson
The Portfolio Team
Faircourt Asset Management
Sub-Advisor to the Ninepoint Alternative Health Fund
Ninepoint Alternative Health Fund - Compounded Returns¹ as of June 30, 2023 (Series F NPP5421) | Inception Date - August 8, 2017
| MTD | YTD | 3MTH | 6MTH | 1YR | 3YR | 5YR | INCEPTION (ANNUALIZED) |
|
|---|---|---|---|---|---|---|---|---|
| FUND | -0.5% | -11.7% | -3.2% | -11.7% | -18.7% | -8.9% | -5.7% | 3.3% |
| TR CAN/US HEALTH CARE BLENDED INDEX | -1.1% | 5.3% | 1.5% | 5.3% | -1.7% | -12.9% | -14.3% | -6.7% |
Statistical Analysis
| FUND | TR CAN/US HEALTH CARE BLENDED INDEX | |
|---|---|---|
| Cumulative Returns | 21.5% | -33.8% |
| Standard Deviation | 27.6% | 29.4% |
| Sharpe Ratio | 0.1 | -0.3 |
1 All returns and fund details are a) based on Series F units; b) net of fees; c) annualized if period is greater than one year; d) as at June 30, 2023. The index is 70% Thomson Reuters Canada Health Care Total Return Index and 30% Thomson Reuters United States Healthcare Total Return Index and is computed by Ninepoint Partners LP based on publicly available index information.
The Fund is generally exposed to the following risks. See the prospectus of the Fund for a description of these risks: Cannabis sector risk; Concentration risk; Currency risk; Cybersecurity risk; Derivatives risk; Exchange traded fund risk; Foreign investment risk; Inflation risk; Market risk; Regulatory risk; Securities lending, repurchase and reverse repurchase transactions risk; Series risk; Specific issuer risk; Sub-adviser risk; Tax risk.
Ninepoint Partners LP is the investment manager to the Ninepoint Funds (collectively, the “Funds”). Commissions, trailing commissions, management fees, performance fees (if any), and other expenses all may be associated with investing in the Funds. Please read the prospectus carefully before investing. The indicated rate of return for series F shares of the Fund for the period ended June 30, 2023 is based on the historical annual compounded total return including changes in share value and reinvestment of all distributions and does not take into account sales, redemption, distribution or optional charges or income taxes payable by any unitholder that would have reduced returns. Mutual funds are not guaranteed, their values change frequently and past performance may not be repeated. The information contained herein does not constitute an offer or solicitation by anyone in the United States or in any other jurisdiction in which such an offer or solicitation is not authorized or to any person to whom it is unlawful to make such an offer or solicitation. Prospective investors who are not resident in Canada should contact their financial advisor to determine whether securities of the Fund may be lawfully sold in their jurisdiction.
The opinions, estimates and projections (“information”) contained within this report are solely those of Ninepoint Partners LP and are subject to change without notice. Ninepoint Partners makes every effort to ensure that the information has been derived from sources believed to be reliable and accurate. However, Ninepoint Partners assumes no responsibility for any losses or damages, whether direct or indirect, which arise out of the use of this information. Ninepoint Partners is not under any obligation to update or keep current the information contained herein. The information should not be regarded by recipients as a substitute for the exercise of their own judgment. Please contact your own personal advisor on your particular circumstances. Views expressed regarding a particular company, security, industry or market sector should not be considered an indication of trading intent of any investment funds managed by Ninepoint Partners. Any reference to a particular company is for illustrative purposes only and should not to be considered as investment advice or a recommendation to buy or sell nor should it be considered as an indication of how the portfolio of any investment fund managed by Ninepoint Partners is or will be invested. Ninepoint Partners LP and/or its affiliates may collectively beneficially own/control 1% or more of any class of the equity securities of the issuers mentioned in this report. Ninepoint Partners LP and/or its affiliates may hold short position in any class of the equity securities of the issuers mentioned in this report. During the preceding 12 months, Ninepoint Partners LP and/or its affiliates may have received remuneration other than normal course investment advisory or trade execution services from the issuers mentioned in this report.
Ninepoint Partners LP: Toll Free: 1.866.299.9906. DEALER SERVICES: CIBC Mellon GSSC Record Keeping Services: Toll Free: 1.877.358.0540
Related Funds
Historical Commentary
- Alternative Health Fund 12/2023
- Alternative Health Fund 11/2023
- Alternative Health Fund 10/2023
- Alternative Health Fund 09/2023
- Alternative Health Fund 08/2023
- Alternative Health Fund 07/2023
- Alternative Health Fund 05/2023
- Alternative Health Fund 04/2023
- Alternative Health Fund 03/2023
- Alternative Health Fund 02/2023
- Alternative Health Fund 01/2023



